|
|

|
General Physics Lab: Heat and Temperature
|
Return to Physics Labs
Equipment and setup
- Equipments used in this experiment are (see Figure 1): Stainless steel
temperature probe, metal cylinder and block, styrofoam cup, calorimeter
cup, 100 ml beaker, balance, metal beaker, hot plate, mercury thermometer,
paper towels, and ice.
flags, two photogates, four 0.05kg masses.
- Turn the setting of the hot plate to "high" to speed up the
heating (see Figure 2).
- Do not let the probe touch the hot metal sample (see Figure 3).
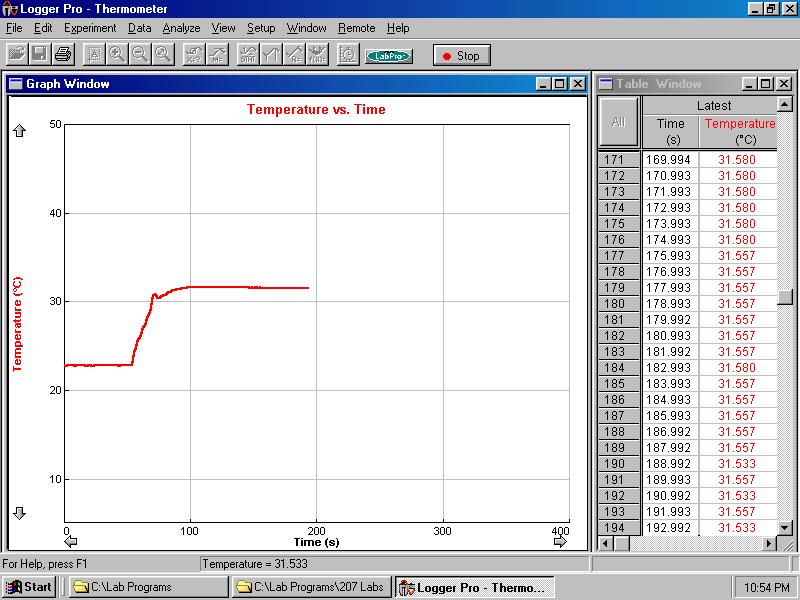
- Use 'Lab programs -> 207 Labs -> Thermometer' program to record
the data. Take data for about one minute to have an accurate measurement
of the initial water temperature.(see Figure 4).
|
[Click on images to enlarge.]
|
TA Notes
- Perform the experiment ahead of time!
- Don't let the temperature probe touch the hot metal samples (Figure 3).
- For you and other's safety, don't touch the the metal beaker when it is
being heated.
- Adjust the position of the mercury thermometer so it won't touch either
the beaker or hot metal.
- Let the metal samples stay in boiling water long enough to make sure
their temperature is the same as the one you read from the mercury
thermometer.
- Make sure that the students handle the wires and cables properly so they
are not in contact with the hot plate!
- You may want to remind the students the relation between the calories and
joules.
- Double check to make sure all the hot plates have been turned off before
you leave the room!
Data, Results and Graphs
Enter TA password to view sample data and results of this
experiment:
Lab Manual
The Lab Manual for this experiment does not exist on-line.
CUPOL Experiments
Take a look at these CUPOL experiments for an on-line laboratory
experience:
If you have a question or comment, send an e-mail to Lab Coordiantor:
Jerry Hester
Return to Physics Labs
|
|